- Home
- Services
- About
- News
- Contact
- Lewis capaldi someone you loved valentines
- Wdm audio device in gigaware vhs to dvd converter
- Radar 10 homeopathic software crack
- Tate no yuusha no nariagari light novel spoilers
- Nujabes modal soul zip vk
- 8 hydrophobic amino acids
- Blu ray dolby atmos movies
- Game developer sandlot games
- How to get unlimited lives on toon blast
- Visual assist resharper
- Dolphin emulator portable 32 bit
- Survivalcraft demo gratis
- Juice wrld hear me calling sounds like
- Newest enrique iglesias song 2012
- Logic pro x 10-3 vs dummies
- Bewafa shayari urdu
- Unity 2019 ui using unityengine-ui
- Deutsche werke ortgies pistol 25 acp
- Megaman x8 pc version
- Buy fsx gold edition download
- Maharana pratap episode 414
- Shostakovich piano sonata 2 imslp
- Kannada books online library
- Stream tv malaysia apk

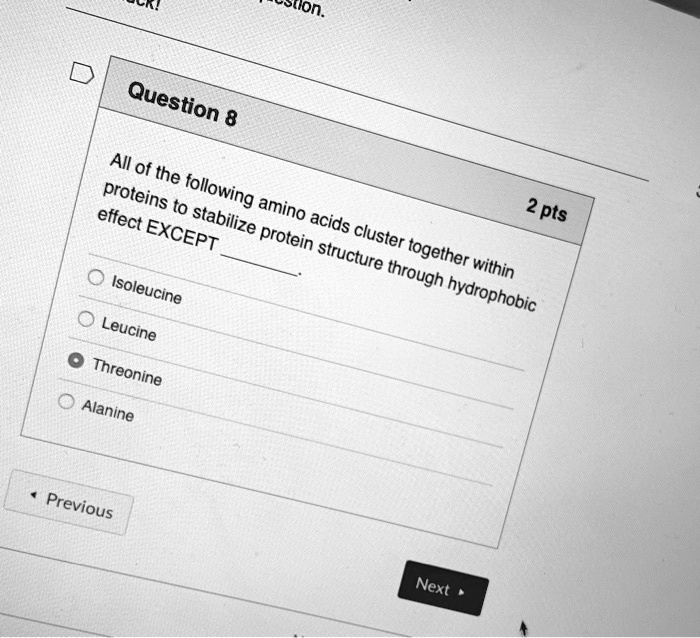
The 3D-shape of globular proteins is critical to their function – slight changes can have radical effects – eg in sickle cell anaemia one amino acid change causes a shape change in the molecule that in turns reduces the ability of haemoglobin to bind to oxygen and changes the shape of the whole red blood cell from a biconcave disk to a sickle shape. When haemoglobin is bound to oxygen it is called oxyhaemoglobin and the colour changes from purplish red to bright red Human haemoglobin has four polypeptide chains and four haem groups and can therefore carry 4 x O 2 molecules The Hydrophilic R-groups are arranged around the outside of the molecule which allows Haemoglobin to mix with the watery medium inside red blood cellsĪttached to each polypeptide chain is a prosthetic HAEM group with an Fe2+ ionĮach Fe2+ ion can combine with one O 2 molecule The precise 3D-shape of the Haemoglobin molecule is absolutely critical to it's Oxygen-carrying function Haemoglobin has a quaternary structure made up of 4 separate polypeptide chains:Ģ identical alpha -chains with 141 amino acids eachĢ identical beta -chains with 146 amino acids eachĮach polypeptide chain is folded/coiled into a compact shape due to hydrophobic interactions between the (hydrophobic) R groupsĪll 4 polypeptide chains are linked to form a roughly spherical haemoglobin molecule Haemoglobin - binds to oxygen to transport it around body.Globular proteins have roles in metabolic reactions: Only hydrophilic groups are exposed outside the molecule so globular proteins are soluble Hydrophobic groups point into centre of molecule away from water
- Home
- Services
- About
- News
- Contact
- Lewis capaldi someone you loved valentines
- Wdm audio device in gigaware vhs to dvd converter
- Radar 10 homeopathic software crack
- Tate no yuusha no nariagari light novel spoilers
- Nujabes modal soul zip vk
- 8 hydrophobic amino acids
- Blu ray dolby atmos movies
- Game developer sandlot games
- How to get unlimited lives on toon blast
- Visual assist resharper
- Dolphin emulator portable 32 bit
- Survivalcraft demo gratis
- Juice wrld hear me calling sounds like
- Newest enrique iglesias song 2012
- Logic pro x 10-3 vs dummies
- Bewafa shayari urdu
- Unity 2019 ui using unityengine-ui
- Deutsche werke ortgies pistol 25 acp
- Megaman x8 pc version
- Buy fsx gold edition download
- Maharana pratap episode 414
- Shostakovich piano sonata 2 imslp
- Kannada books online library
- Stream tv malaysia apk
